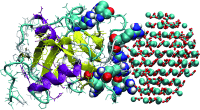
Eggshells of chickens are formed from amorphous calcium carbonate nanoparticles which are transformed into ordered crystals by protein mediation.
Eggshell this a thin yet relatively strong material due to its composite makeup of ordered calcium carbonate crystals and . A layer of crystalline calcite clusters overlays partially aligned calcite columns. The mineral begins as amorphous calcium carbonate nanoparticles, then is transformed to ordered crystals by the presence of C-type lectin-type proteins. The proteins attach themselves to the nanoparticles, initiating crystal transformation, and then detach while crystal growth continues.