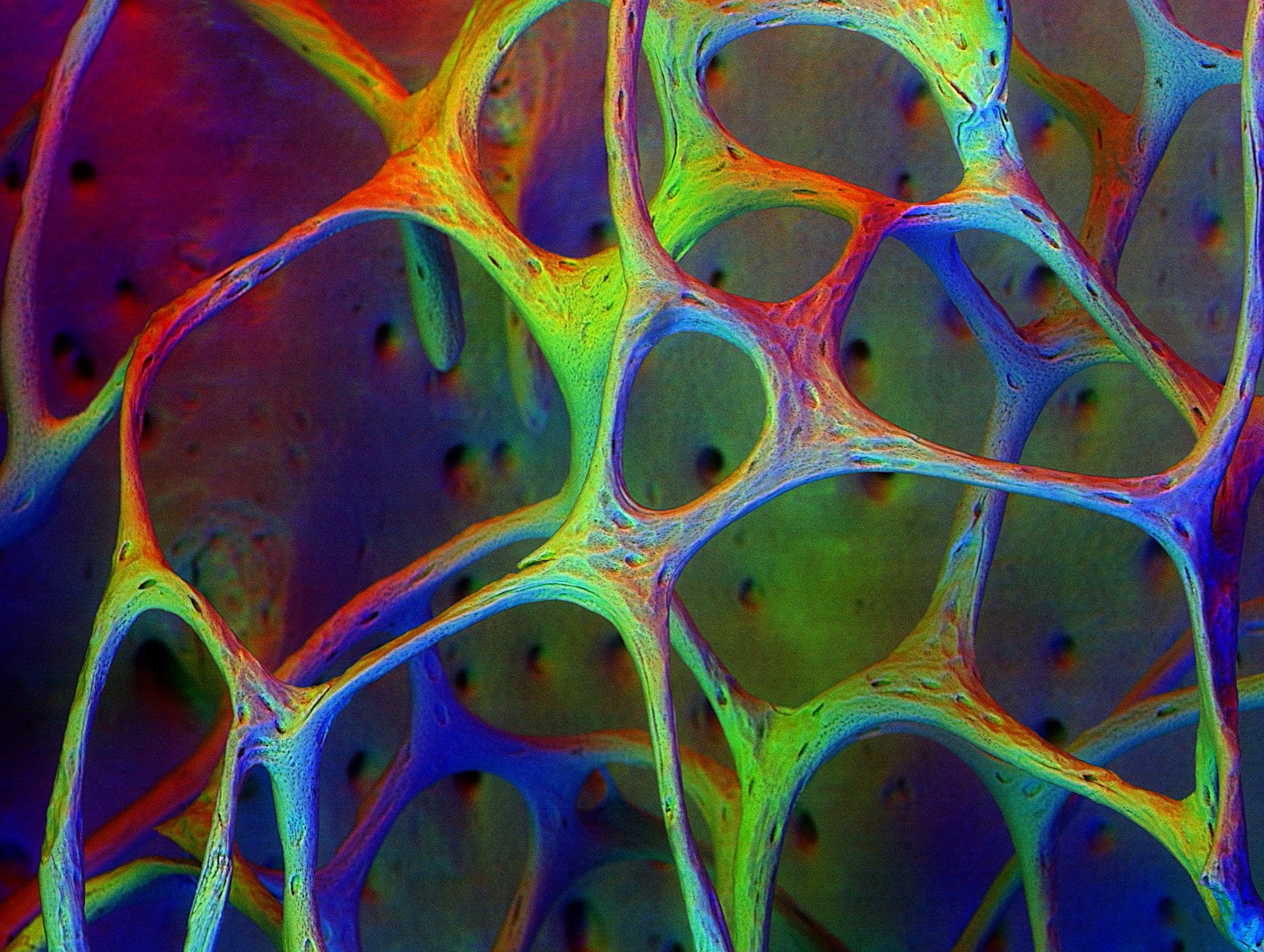
A combination of mineral crystals and collagen fibers protects bone from major fractures by sacrificing small structural elements.
Introduction
In our hands, in our legs, around our brains: Bones provide structural support for the bodies of humans and of every other vertebrate, from fish to tigers to birds.
It might seem that bone is inflexible, but its basic component is a springy and versatile called collagen––found in elastic tissues like skin, tendons, and ligaments. Collagen molecules consist of three intertwining spiral chains, allowing the molecules themselves to bend and stretch. Individual molecules then overlay one another in a staggered pattern like bricks in a wall, forming relatively fine fiber structures called “fibrils.” If bone were comprised of just collagen, it might make yoga poses easier but it wouldn’t be very strong.
The Strategy
Enter calcium, the mineral that gives bone its strength and hardness. When bone develops, calcium crystals form in the gaps and bond to the flexible collagen like mortar amid the bricks.
Dr. Markus Buehler, a materials engineering professor at the Massachusetts Institute of Technology, compared the stress behavior of ordinary collagen fibrils to mineralized collagen fibrils. Buehler analyzed the stress exerted on bundles of fibrils under the strain of pulling their two ends apart. At first, fibrils with and without minerals both stretch like a rubber band. They don’t stretch as much as a rubber band, but the stress on the tissue increases linearly with the strain of pulling. Keep tugging, however, and the fibrils deform, releasing the tension the way caramel candy does when you stretch it.
This is the point where the calcium makes a difference. Pull even harder on pure collagen fibrils and they continue to behave like caramel candy. During what’s called plastic deformation, molecules easily slip past one another and only a tenuous string connects the two ends. It becomes easier and easier to pull them apart as the wispy connective string stretches and thins. Eventually, failure occurs and the two ends separate.
The stress lurches down, then up again as the strong bonds between crystals and collagen break one at a time. This spreads the molecular slipping over many stages.
In contrast, when mineralized collagen fibrils begin plastic deformation, their slipping molecules are quickly interrupted. The stress lurches down, then up again as the strong bonds between crystals and collagen break one at a time. This spreads the molecular slipping over many stages. Imagine trying to open an extension ladder that catches on every rung. Tugging hard cracks the bond between one mineral and one collagen molecule and releases a single ladder rung. But then the ladder snags on another bond until enough force breaks that and pulls the next rung down. Before reaching structural failure, all the bonds must break. In this way, minerals dissipate energy, permitting failure on minute scales to protect the entire bone from fracturing.
The Potential
Bones need to be tough but able to dissipate the forces we put them under. If they couldn’t bend slightly, they’d snap apart every time we jumped or fell. In that regard, bones are like other composite building materials that give man-made structures strength while imparting the flexibility to dissipate energy from wind, earthquakes, and storms. Furthermore, engineering design—like staged molecular slipping—usually includes redundancy so that when small elements fail, the overall structure remains intact.